|
For instance, water boiling, wax melting, wood chopping, dissolving sugar and more. In other words, the original characteristics of the substance will not change. 
The same element or compound will exist prior to or after the change. All this happens without a change in molecular composition. In this type of change, the substance will experience a change in its physical properties like shape, size, volume and more. This impacts the chemical properties as well as the physical propertiesĭuring the reaction, absorption and evolutionĬutting of wood, freezing water, melting ice cubes, crushing a tomato are physical changesīurning of wood, digesting of food, baking of cake, rusting of iron are examples It impacts the physical properties like shape, size, colour and more This is a change where the molecules rearrange themselves but no changes in internal composition happenĬhemical change transforms the substance into a new one and gives it a different chemical composition Important Difference Between Physical And Chemical Change Basis of Comparison Difference Between Physical And Chemical Change So, it is a permanent change which alters the melting point, taste, colour and more. Moreover, these changes are not easily reversible, in fact, they are irreversible. 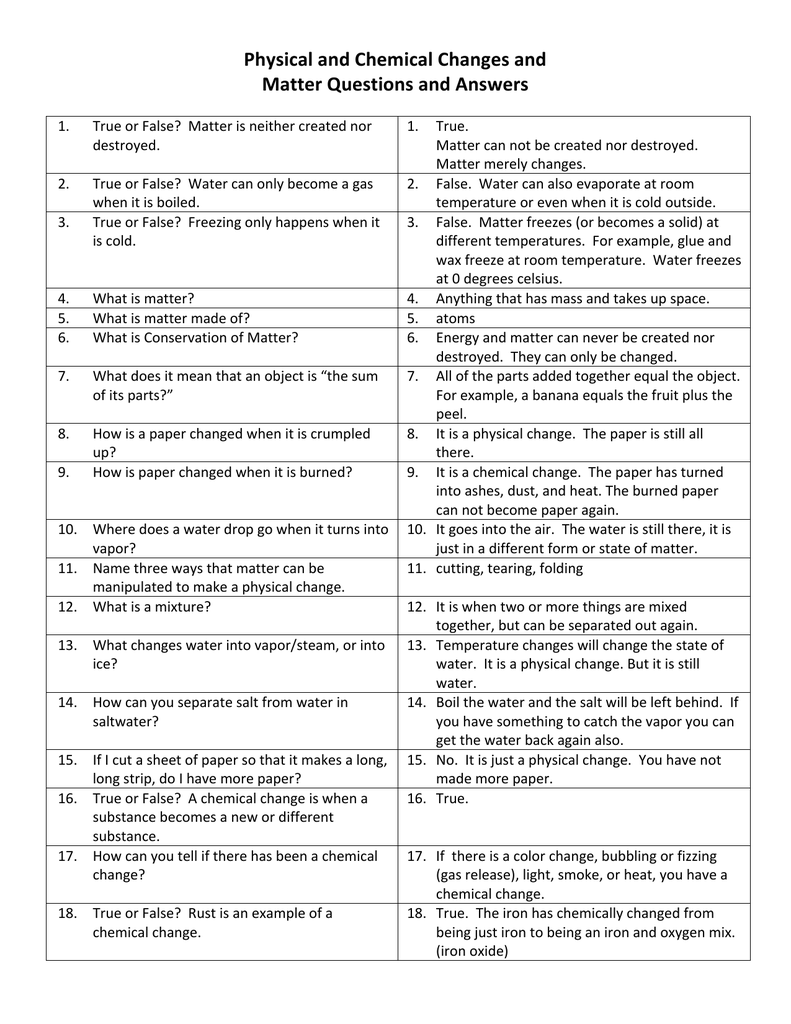
It is because the atoms plus molecules of the substances start rearranging themselves to form a new product. In other words, chemical changes do alter the properties of a substance throughout the reaction. Thus, there is a change in the composition of the material forming a new product. For instance, when you burn a piece of paper or wood, it will change into ash. It results in a new product after the reaction. Definition of Chemical ChangeĪ chemical change is the exact opposite of physical change. Moreover, the changes are usually reversible and temporary. A physical change does not bring any alternation of any kind on the chemical properties. Thus, this will help you in understanding the difference between physical and chemical change better. Similarly, when water turns into ice or vapour, the chemical properties remain the same, only the physical state changes. However, the properties will be the same. For instance, when you tear a paper, it will only change its shape and size. In here, the chemical properties do not change. 
Physical change is the kind of change where the matter switches into another form. 3.2.1 FAQs about Physical Change and Chemical Change Definition of Physical Change
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
